Abstract
This narrative review provides an up-to-date overview of atypical femoral fractures (AFFs), emphasizing diagnostic criteria, epidemiology, pathophysiology, risk factors, and evaluation with screening strategies. AFFs are rare but significant complications associated with prolonged bisphosphonate (BP) therapy for osteoporosis. Although the pathogenesis of AFFs has not been fully elucidated, its primary mechanism is thought to involve impaired bone remodeling, leading to unhealed microfractures that progress to stress fractures under repetitive loading. AFFs can occur in various regions of the femur, influenced by femoral geometry and the lower limb axis. Other risk factors include prolonged steroid use, arthroplasty, genetic predispositions, and metabolic bone disorders. The diagnosis of AFFs is based on criteria established by the American Society for Bone and Mineral Research. Key radiographic features include lateral cortical transverse fracture lines and localized cortical thickening, typically with minimal or no comminution on the medial cortex. Dual-energy X-ray absorptiometry for screening tests and magnetic resonance imaging as an advanced imaging modality enable the early detection of incomplete fractures. This multi-modal approach facilitates the prompt identification of prodromal cortical changes, reducing the risk of complete fractures in high-risk populations, particularly patients undergoing prolonged BP therapy.
-
Keywords: Femur, Bone fractures, Diphosphonates, Bisphosphonates, Complications
Introduction
The use of bisphosphonates (BPs) revolutionized osteoporosis treatment, significantly reducing fracture rates and improving patient outcomes [
1]. By suppressing bone turnover, BPs increase bone mineral density; however, prolonged use may lead to atypical femoral fractures (AFFs) due to altered bone biomechanics [
2]. Although the exact mechanism behind the pathogenesis is not yet fully understood, AFFs are stress fractures located between the lesser trochanter and the distal metaphyseal flare, often associated with a negative side-effect of antiresorptive therapy [
3].
In 2005, reports of atypical subtrochanteric femoral fractures began to surface, followed by case series and registry-based studies that identified a correlation between these fractures and prolonged BP therapy. Despite the clear favorable risk-benefit profile, where approximately 1,200 fragility fractures are prevented for every AFF associated with BP therapy, these findings prompted a decline in routine use [
4]. Given the extensive cohort of individuals benefiting from this pharmacological intervention, the incidence of AFF remains comparatively low [
5-
8]. Furthermore, recent studies have identified additional risk factors contributing to the development of AFFs in BP-naive patients [
9,
10].
General management involves discontinuing BP to avoid over-suppression of bone remodeling, yet the optimal transition to alternative therapies such as osteoanabolic agents is debated [
11-
13]. For surgical treatment, intramedullary nailing (IMN) is considered the most effective option, as other methods of surgical fixation are associated with a higher failure rate [
14-
16]. However, AFFs often occur in mechanically challenging environments, such as anterolateral femoral bowing, where IMN may not properly fit the femoral anatomy [
17]. Moreover, due to the absence of randomized controlled trials establishing the optimal treatment for AFFs, management is currently guided by expert opinions and lacks clear, evidence-based consensus.
Given the complexity of AFFs, this review aims to provide an updated overview of AFFs by analyzing current evidence on their definition, pathogenesis, epidemiology, diagnosis, and management, with a focus on key clinical considerations.
Ethics Statement
All procedures and treatment performed involving human participants were in accordance with the ethical standards of the institution. All photos included in this review were obtained with informed consent from the patients for publication.
Definition
AFFs are distinct from typical hip and femoral fractures, characterized by their location, morphology, and association with specific risk factors. The American Society for Bone and Mineral Research (ASBMR) first established diagnostic criteria for AFFs in 2010, which were later updated in 2013 to improve clarity and diagnostic accuracy [
18]. These criteria define AFFs based on major and minor features, requiring the presence of at least four out of five major features for diagnosis (
Table 1) [
18].
Major criteria include fractures that are low-energy or atraumatic, transverse or slightly oblique in orientation, minimally comminuted, and extending through both cortices with a medial spike or, in incomplete cases, confined to the lateral cortex. Lateral cortical thickening is another key feature. Minor criteria are non-essential but often observed, including periosteal thickening, prodromal symptoms like thigh or groin pain, bilateral occurrence, delayed healing, and common comorbidities such as vitamin D deficiency or BP use.
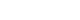
Radiographic features, such as the “dreaded black line” on the lateral cortex, are diagnostically significant, providing insights into fracture behavior and therapeutic strategies. A lateral cortex transverse fracture pattern demonstrated the highest sensitivity, with a mean of 93.6%, and specificity, with a mean of 95.5%, for accurately distinguishing AFFs from other types of femoral fractures (
Fig. 1). Additionally, minimal or absent comminution showed a high sensitivity with a mean of 93.2% [
19].
Epidemiology
The potential association between long-term BP therapy and an increased risk of AFFs was first highlighted in 2005 [
20]. Phase 3 trials for BP approval, conducted from the late 1980s to 2005, did not detect AFFs, as they were not yet defined as a distinct entity. In response, the ASBMR convened a task force to develop consensus definitions in 2010 and 2014. Subsequent to these efforts, further investigations were conducted, resulting in several published studies [
21-
23]. Although AFFs may occur in individuals without prior exposure to BPs, they are most commonly associated with prolonged BP use.
The incidence of AFFs is low, with reported rates varying widely based on study design and population, ranging from 2.3 to 130 cases per 100,000 patient-year [
24,
25]. Dell and Greene [
26] reported the incidence of AFF in the United States and its relationship with BP use, indicating an incidence of 1.8, 38.9, and 107.5 cases per 100,000 patient-years associated with 2, 6 to 8, and greater than 10 years of BP use, respectively. Lo et al. [
27] reported that the risk of AFFs increases with the duration of BP exposure. The age-adjusted incidence rises from 9 per 100,000 person-years for 2 to 4 years of use to 112 per 100,000 person-years after more than 8 years. Similarly, Black et al. [
23] found that the hazard ratio for AFFs increased from 8.9 (95% confidence interval [CI], 2.8–28.2) for 3 to less than 5 years of use to 43.5 (95% CI, 13.7–138.2) for 8 years or more. Furthermore, the incidence of AFFs has increased, likely due to improved screening and diagnostic accuracy. The prevalence of AFFs has doubled over a 7-year period, and it is expected to increase further in the future, probably due to the widespread use of BPs from the current study [
28].
Multiple studies have demonstrated a stronger association of AFFs with Hispanic and Asian descent. One study reported an eightfold higher incidence of AFFs in Asian women compared to White women due to differences in neck-shaft, center edge, and tibiofemoral angles [
27]. Analyzing data from 16 tertiary medical centers in Korea, Lee et al. [
29] reported that among 1,361 cases of proximal femoral fractures, only 1.2% were classified as AFFs. A recent report by Lim et al. [
7] revealed that AFFs accounted for 2.95% of 6,644 hip and femoral fracture cases.
Pathogenesis
The pathogenesis of AFF is not yet fully understood and is considered multifactorial. However, its key characteristics align with the concept of a stress or insufficiency fracture [
18]. Radiographic features such as a general transverse orientation, the lack of comminution, and the localized cortical thickening at the fracture site are indicative of stress fractures [
24]. The primary mechanism of AFF is impaired bone remodeling, where microfractures fail to heal, leading to stress fractures from repetitive loading at specific trigger points on the femoral bone.
Bisphosphonates
Prolonged BP use in patients with AFF inhibits osteoclast activity and bone resorption, while bone formation initially continues, leading to increased bone density and strength. Eventually, both bone formation and resorption are suppressed, resulting in impaired bone remodeling. Clinical and laboratory studies indicate that bone resorption biomarkers remain suppressed for up to 1 year, with evidence suggesting a biological half-life for potent BPs exceeding 10 years following a single dose [
30,
31]. Ahn et al. [
32] reported that the AFF group exhibited lower levels of C-terminal telopeptide of type I collagen, indicating more suppressed bone remodeling, which may be associated with the occurrence of AFFs. While BPs effectively reduce the risk of fragility fractures in patients with osteoporosis, their prolonged use is associated with an increased risk of AFFs. Black et al. [
33] reported a sixfold increase in the risk of atypical hip fractures after 5 to 8 years of BP therapy and a 13.1-fold increase following more than 8 years of use.
The pathogenesis of AFFs associated with prolonged BP use remains incompletely defined, though proposed mechanisms include reduced bone turnover, compromised microdamage repair, and alterations in bone quality [
2,
18,
34]. Studies have shown a reduced fatigue life of cortical bone after alendronate treatment without changes in mineralization, along with findings of widened Haversian canals and diffuse microdamage in biopsy specimens from BP-related AFFs, suggesting increased stress concentration and impaired microdamage repair [
35]. However, other research has indicated no significant differences in bone microarchitecture between patients with AFFs taking BPs and controls, implying that additional factors may contribute to these fractures [
36].
Femoral Morphology
From a biomechanical perspective, the medial femoral cortex always experiences compressive forces, while the lateral femoral cortex is subjected to tensile forces. Alterations in femoral geometry and the mechanical axis of the lower extremities can exacerbate tensile forces on the lateral cortex [
36]. Notably, increased lateral and anterior bowing of the femur has been strongly correlated with elevated mechanical strain on the femur, particularly at sites vulnerable to AFFs [
37]. Several studies have demonstrated that anterolateral femoral bowing, varus alignment of the lower limb, a reduced neck-shaft angle, and excessive femoral offset contribute to stress concentration at sites predisposed to AFFs [
37-
46]. Atypical femoral shaft fractures exhibit distinct geometric features, including a lower radius of curvature, greater femur width, and longer anteroposterior length, whereas subtrochanteric AFFs show no significant geometric differences. These findings highlight the role of altered femoral geometry in shaft AFFs [
47]. Computed tomography (CT)-based finite element analysis identified the femoral weakest point under tensile stress and its association with lower limb geometry. The results matched AFF locations and showed a distal shift with varus knee alignment, highlighting the influence of limb axis and femoral bowing in AFF development [
48].
Genetics
Recent studies emphasize the complex interplay of genetic susceptibility, mechanical stress, and antiresorptive therapy. Genetic variants in key genes, such as
SLC34A1,
SLC9A3R1,
GGPS1, and
CYP1A1, have been identified as potential contributors to AFFs [
49-
52]. These genes are involved in pathways regulating bone mineralization, osteoclast function, and drug metabolism, suggesting a heightened vulnerability in patients with these variants.
Comprehensive genetic analyses, including whole-exome sequencing and gene panel studies, have highlighted the polygenic nature of AFFs [
51,
53,
54]. Variants in genes such as
PLOD2,
XRN2,
SUCO, and members of the Wnt signaling pathway such as
LRP5,
WNT1 were recurrently observed. Furthermore, monogenic bone disorders like osteogenesis imperfecta and hypophosphatasia were linked to AFF in subsets of patients, with pathogenic variants detected in about 15%–33% of individuals in various cohorts [
50,
53,
54].
While the precise genetic and environmental interactions remain unclear, these findings underscore the importance of personalized approaches to therapy. Identifying at-risk patients through genetic screening could improve AFF prevention strategies, particularly in those receiving long-term antiresorptive treatment. However, the variability in genetic findings emphasizes the need for larger, more diverse studies to fully clarify the genetic factors contributing to AFF.
Denosumab
Denosumab, an alternative antiresorptive therapy, has shown a possibility of AFF occurrence, as bilateral AFFs have been observed in a patient undergoing denosumab treatment [
55]. In the FREEDOM (Fracture Reduction Evaluation of Denosumab in Osteoporosis Every 6 Months) trial and its extension, conducted between 2004 and 2018 with 7,808 enrolled patients (4,550 in the extension phase), only one AFF was reported in each group (long-term and crossover) during up to 10 years of denosumab treatment [
56]. In addition, several studies reported a very low incidence of AFFs despite prolonged exposure to denosumab [
10,
57,
58]. Current evidence suggests that the risk of AFFs associated with denosumab is very low. Considering its favorable risk-benefit profile, denosumab remains a viable and potentially safer option for osteoporosis treatment compared to BPs. However, it is essential to reassess the ideal duration of antiresorptive therapy and carefully evaluate the safety of transitioning between treatments, such as BPs and denosumab, to optimize patient outcomes.
Total Joint Arthroplasty and Implant
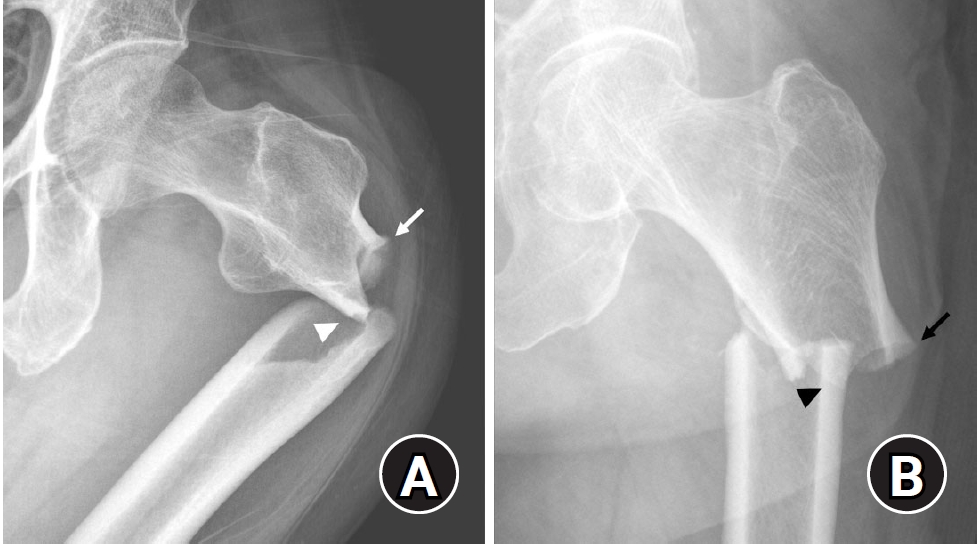
BP use is strongly linked to atypical periprosthetic femoral fractures (APFFs), which account for 8%–10% of periprosthetic fractures and are increasingly observed, particularly in patients with prolonged BP exposure (
Fig. 2) [
59,
60].
AFFs associated with arthroplasty and implants, including peri-implant and periprosthetic fractures, have distinct characteristics and pathophysiological mechanisms. Lee et al. [
61] reported that 10.4% of Vancouver type B periprosthetic femoral fractures were classified as atypical among 67 patients. Kim et al. [
62] identified peri-implant AFFs in 11 elderly women who previously underwent osteosynthesis with plates or nails. These fractures occurred at stress riser sites such as screw holes and were associated with excessive femoral bowing and BP use.
Ferreira et al. [
63] conducted a systematic review and meta-analysis, showing that long-term BP use significantly increases the risk of APFFs, with a relative risk of 14.1. These fractures, which often present with prodromal pain and occur near prosthetic components, are likely stress fractures influenced by implant-induced biomechanical changes. Despite the risks, BPs are still recommended for osteoporosis management, emphasizing the need for clinicians to monitor high-risk patients closely [
63].
Hashimoto et al. [
64] focused on periprosthetic AFFs associated with total knee arthroplasty (TKA). Their prospective multicenter study reported that 6.6% of AFFs were periprosthetic, predominantly in elderly women. Risk factors included femoral bowing, long-term BP exposure, and altered lower limb alignment after TKA. Biomechanical analyses revealed stress concentration at fracture sites, supporting mechanical and biological contributions to fracture development. Regular monitoring with full-length femoral X-rays post-TKA is recommended to detect early fracture signs [
64].
Other Risk Factors
The risk factors for AFFs vary across studies and continue to be actively investigated. Kimura et al. [
65] investigated latent metabolic bone diseases and skeletal dysplasia in 38 Japanese patients with morphologic AFFs over a 10-year period. Among these, 10 patients had a history of glucocorticoid use, 18 had used antiresorptive agents, and four were diagnosed or suspected to have conditions affecting bone formation, including hypophosphatemic osteomalacia, pycnodysostosis, and a genetic variant in alkaline phosphatase, biomineralization associated gene, which encodes the tissue-nonspecific alkaline phosphatase [
65].
Furthermore, Asian ethnicity is a strong independent risk factor for AFF, unaccounted for by ethno-specific differences in proximal femoral geometry. BP use may be associated with a greater predisposition for AFF in Asian subjects compared with non-Asian subjects [
66].
In addition, long-term glucocorticoid use is a significant risk factor for AFFs. Koh et al. [
67] reported that prolonged BP use, long-term glucocorticoid use, and higher BMI increased AFF risk, with hazard ratios of 5.2, 3.0, and 1.2 per 1 kg/m² BMI increase, respectively. Among 35,104 BP users, 43 (0.12%) experienced AFFs after a mean BP exposure of 7.3 years. Similarly, Sato et al. [
68] observed a 26% prevalence of LPT and AFF in five of 121 Japanese patients with autoimmune rheumatic diseases on long-term BP and glucocorticoid therapy, highlighting challenges in managing osteoporosis in this group.
Evaluation and Diagnosis
As the symptoms and signs of AFFs are often mild or nonspecific, maintaining a strong clinical suspicion and performing additional evaluations can enhance diagnostic accuracy. A comprehensive history and thorough clinical assessment are critical in determining the need for advanced imaging studies. However, these symptoms are frequently misinterpreted or attributed to other conditions, such as degenerative spine-related neuropathy or arthritis, potentially delaying proper diagnosis [
69].
Clinical Symptoms and Presentation
Approximately 86% of patients with BP-related AFFs experience prodromal symptoms, typically described as dull, aching pain in the thigh or groin, lasting for weeks or even months [
69]. Other studies have reported that 77% of patients experienced prodromal pain, with a mean duration of 9 months before the fracture occurred [
70]. However, the onset and duration of prodromal pain vary widely, ranging from a single day to several years [
71]. This pain is typically localized and unrelated to significant trauma, distinguishing it from other musculoskeletal injuries. A detailed patient history is crucial, particularly regarding the duration of BP use and coexisting risk factors such as glucocorticoid therapy. These symptoms warrant further investigation, as they may indicate stress reactions or incomplete fractures. Early recognition of these warning signs is essential to prompt timely imaging and intervention to prevent fracture progression.
Image Study
Imaging modalities primarily consist of plain radiographs, which should cover the pelvis, full-length bilateral femurs, and a scanogram to assess lower limb alignment. The radiologic characteristics of AFF include a transverse fracture line originating from the lateral cortex that extends transversely or in a short oblique direction toward the medial cortex. Complete fractures typically involve both cortices and may present with a medial spike, while incomplete fractures are confined to the lateral cortex. Fracture comminution is minimal or absent. Localized periosteal or endosteal thickening of the lateral cortex, often described as "beaking" or "flaring" is a key feature, along with cortical thickening of the femoral diaphysis. These findings are essential for accurate diagnosis and assessment on imaging studies [
18].
Patients presenting with prodromal symptoms but negative radiologic findings, or asymptomatic individuals with high clinical suspicion, should undergo further evaluation using advanced imaging techniques such as CT, magnetic resonance imaging (MRI), or a technetium-99 bone scan (
Fig. 3). MRI plays a crucial role in identifying stress fractures, including AFFs and other less common types, ensuring timely diagnosis and appropriate treatment [
72]. A study proposed an MRI-based scoring system to predict symptoms and fracture risk in BP-related femoral periosteal stress reactions (
Fig. 4). While MRI and combined scores were linked to symptoms, only the radiographic survival score effectively predicted fracture risk [
73].
Screening
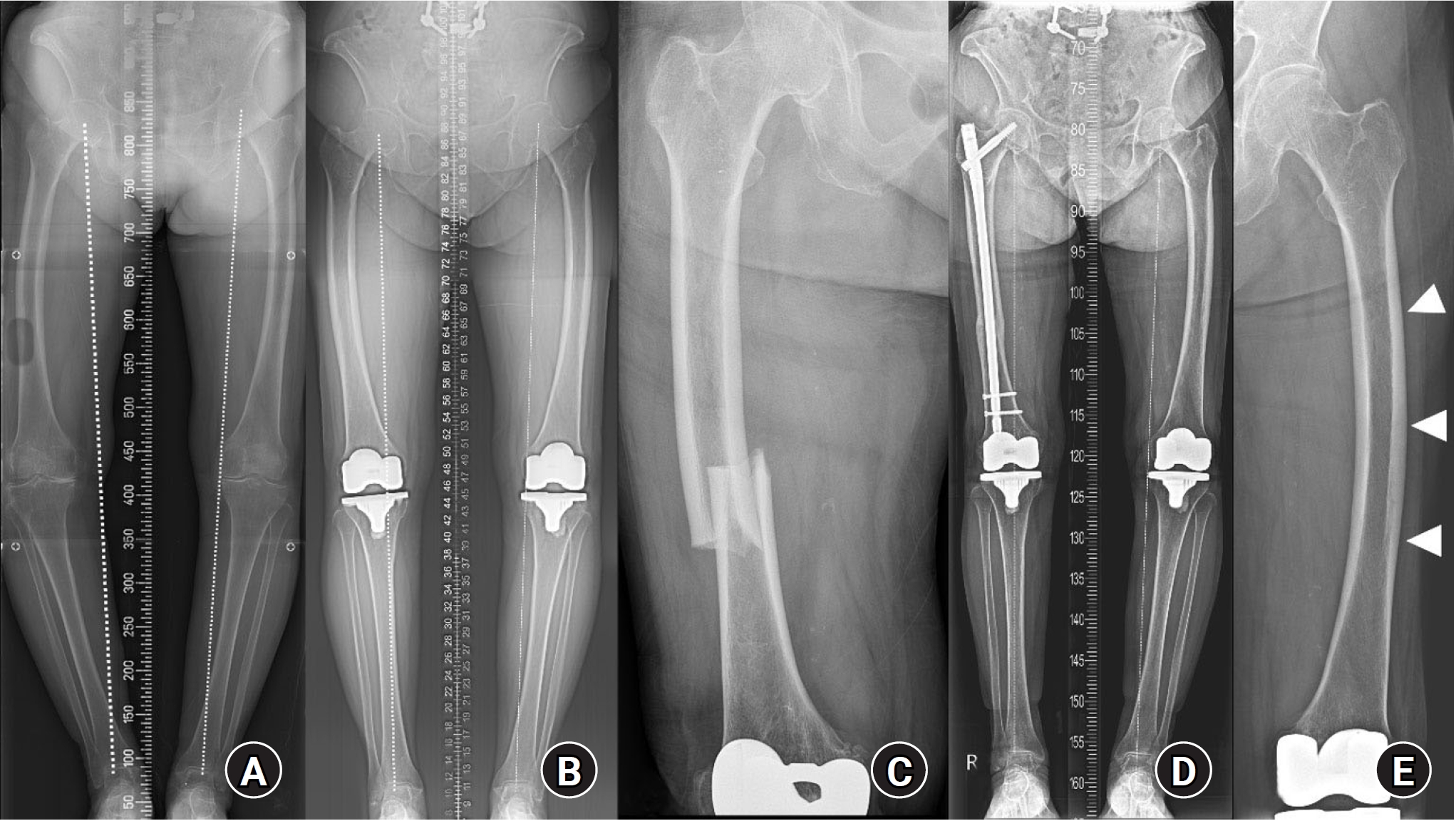
Dual-energy X-ray absorptiometry (DXA) scans, particularly extended femur imaging, have emerged as effective tools for opportunistic screening of incomplete AFFs (
Fig. 5) [
74]. Cheung et al. [
75] emphasized the importance of full-length femur imaging (FFI) for detecting abnormalities in the spectrum of AFFs, including periosteal and endosteal thickening. Their work highlighted the utility of FFI in patients on prolonged antiresorptive therapy, particularly those undergoing BP or denosumab treatment for over 3 years. They also recommended visual inspection of femoral DXA images to identify focal cortical abnormalities, which can aid in the early identification of incomplete AFFs [
75]. Smith et al. [
76] further supported the role of DXA in opportunistic screening by evaluating the precision of extended femur imaging in measuring cortical abnormalities. Their study demonstrated that extended femur scans accurately detect lateral cortical thickening and “beaking,” a key prodromal feature of AFFs [
76]. The findings suggested the integration of extended femur imaging into routine DXA scanning protocols, especially for monitoring patients at high risk for AFFs. In addition to DXA, advanced imaging modalities such as whole-body scintigraphy (WBS) and positron emission tomography (PET)-CT have shown promise for early detection. Cha et al. [
77] demonstrated that these techniques identified AFF-related lesions months before they became apparent on standard radiographs. Their retrospective study of breast cancer patients receiving high-dose BP therapy revealed that WBS and PET-CT effectively detected prodromal changes, enabling preventive interventions like prophylactic fixation before fractures occurred.
The integration of DXA-based femoral imaging, supplemented with advanced modalities such as WBS and PET-CT, offers a comprehensive approach to early AFF detection. This multi-modal strategy enhances the ability to identify prodromal cortical changes and reduce the risk of complete fractures in high-risk populations, including those on prolonged BP therapy.
Conclusions
AFFs are rare stress fractures influenced by femoral geometry, genetic predispositions, and prolonged use of antiresorptive therapies such as BPs. Risk factors include glucocorticoid use, metabolic bone disorders, and arthroplasty or prosthetic implants, which can contribute to stress fractures in vulnerable regions by increasing mechanical strain due to altered femoral geometry. AFFs often present with prodromal symptoms, such as dull, localized thigh or groin pain, lasting weeks to months, but can be easily misdiagnosed. Imaging plays a critical role in diagnosis, with radiographs localized periosteal or endosteal thickening of the lateral cortex, often described as "beaking" or "flaring." Advanced modalities like MRI, CT, and technetium-99 bone scans enhance the detection of incomplete fractures, while DXA with extended femur imaging is effective for screening. Opportunistic screening and tools like WBS and PET-CT detect early cortical changes, aiding prevention. Integrating these imaging techniques allows timely diagnosis and intervention, reducing the risk of complete fractures in high-risk patients. Future research should focus on identifying genetic markers and biomechanical factors contributing to AFF development to facilitate personalized treatment strategies. Longitudinal studies are needed to evaluate optimal therapy durations and safe transitions between antiresorptive agents. Additionally, investigating the efficacy of advanced imaging techniques and screening modalities in detecting and managing AFFs will provide valuable insights for improving patient outcomes.
Article Information
-
Author contribution
Data curation: JTK. Formal analysis: WTC. Methodology: JHK. Project administration: WTC. Resources: WTC, JTK. Validation: SS. Visualization: WTC, JHK, JTK. Writing - original draft: WTC, JHK, JTK. Writing - review & editing: WTC, SS. All authors read and approved the final manuscript.
-
Conflict of interest
None.
-
Funding
None.
-
Data availability
Not applicable.
Fig. 1.(A) A 73-year-old female patient diagnosed with a subtrochanteric atypical femoral fracture (AFF) exhibited characteristic features, including a transverse fracture line on the lateral cortex (arrow) followed by a short oblique fracture pattern on the medial cortex (arrowhead). (B) A 72-year-old female patient with a subtrochanteric AFF demonstrated similar features, with a transverse fracture line on the lateral cortex (arrow) and an oblique fracture pattern on the medial cortex (arrowhead). In both patients, the medial spike and lateral cortex flaring or beaking was observed.
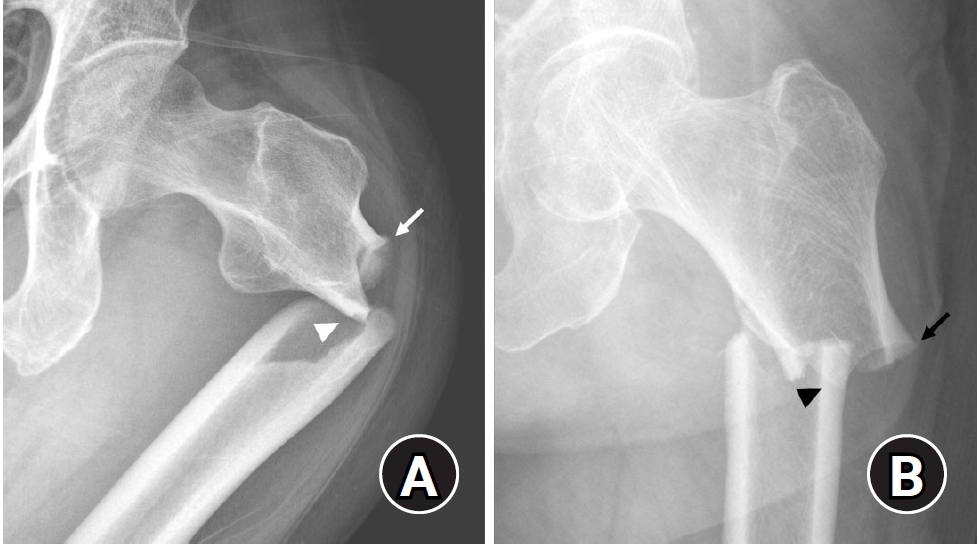
Fig. 2.(A) An 80-year-old female patient with a 6-year history of alendronate use presented with severe varus alignment and degenerative arthritis in both knee joints. (B) Total knee arthroplasty was performed, but varus alignment persisted, indicating grade II anterolateral bowing [
59,
60]. (C) Six years after surgery, a complete atypical femoral fracture (AFF) with characteristic features occurred. (D) Within 1 year of treatment, the AFF healed, accompanied by noticeable improvements in varus alignment and generalized thickening compared to the preoperative state. (E) On the contralateral side (arrowheads), prominent generalized thickening remained evident.
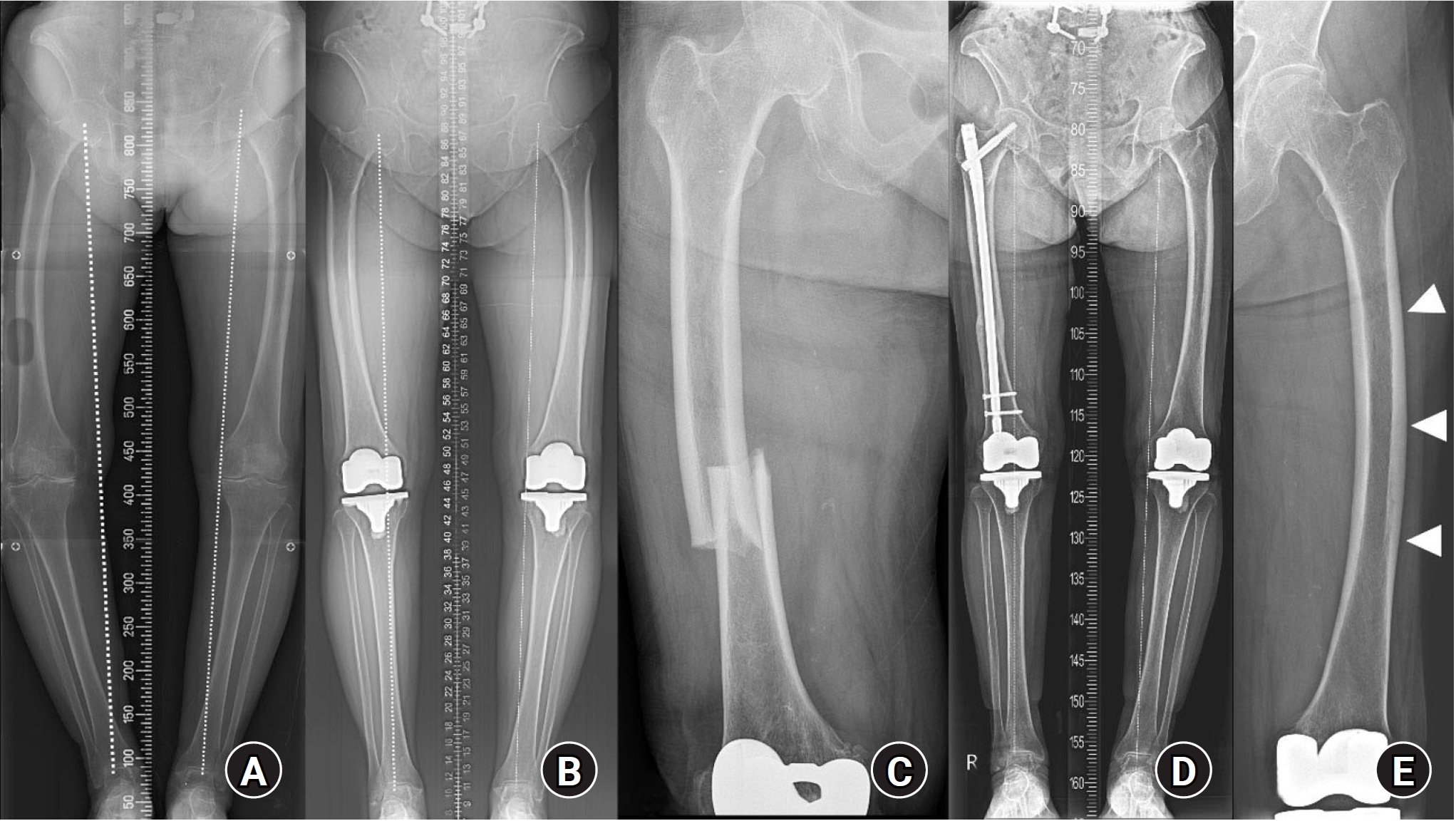
Fig. 3.(A) An 80-year-old female patient with a 10-year history of alendronate use presented with bilateral thigh pain lasting several months. A scanogram showed lower limb varus alignment with coxa vara. (B) Lateral cortex flaring or beaking (arrowhead) and generalized cortical thickening of the femoral shaft were visible, but not prominent. (C) As a further evaluation, a technetium-99 bone scan revealed increased uptake in the left mid-femur (arrows), consistent with atypical femoral fracture (AFF). (D) One month after the initial visit, a complete AFF occurred while preparing for a prophylactic operation.
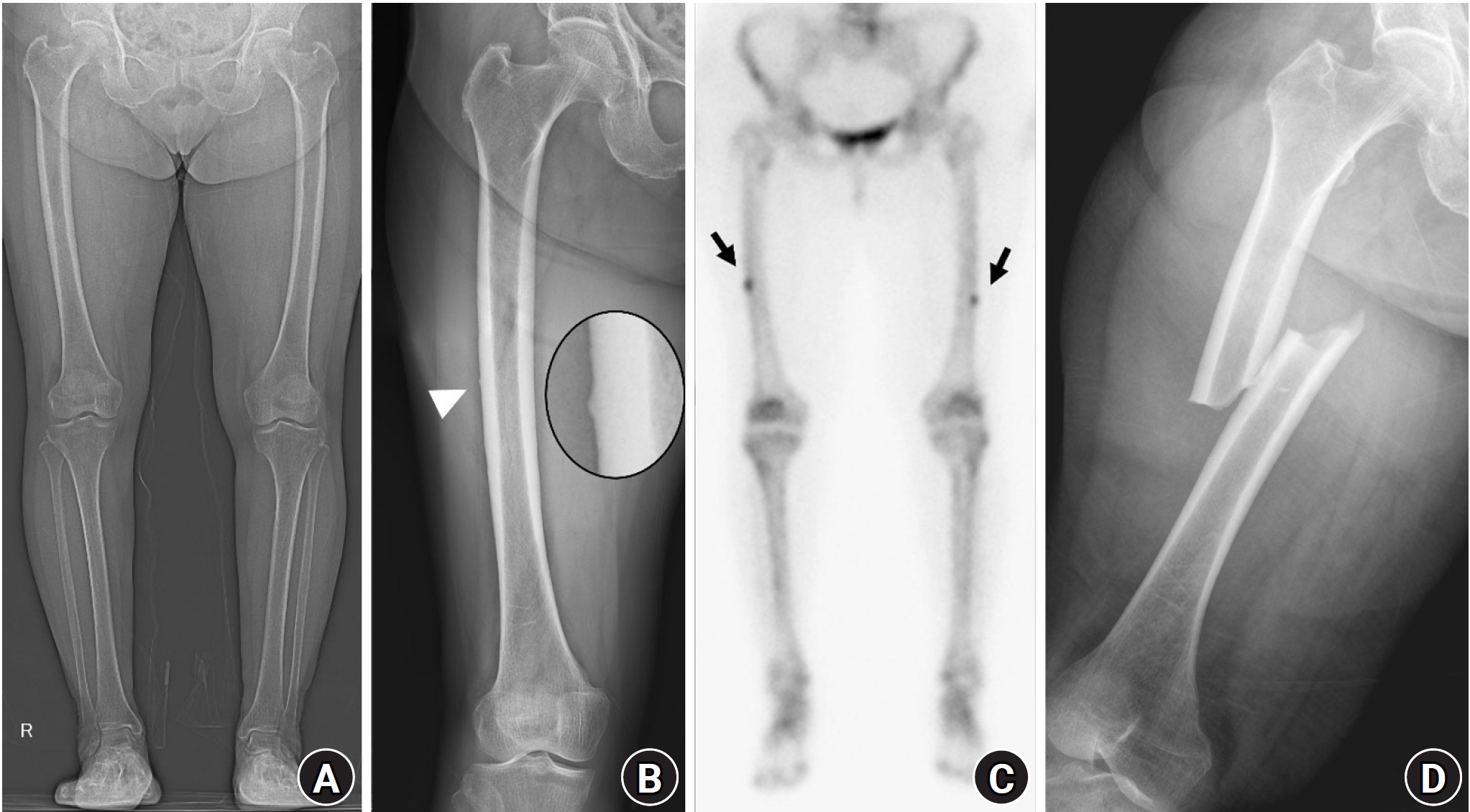
Fig. 4.A 74-year-old female patient with osteoporosis who had been treated with alendronate for 7 years presented with persistent right thigh pain. (A) A lateral cortex lucent fracture line was observed, indicating an incomplete or impending atypical femoral fracture (arrowheads). (B) T2-weighted magnetic resonance imaging showed endosteal edema along with a visible fracture line (arrow). (C) Three years after prophylactic nailing, the lateral cortical beaking and lucent fracture line had diminished.
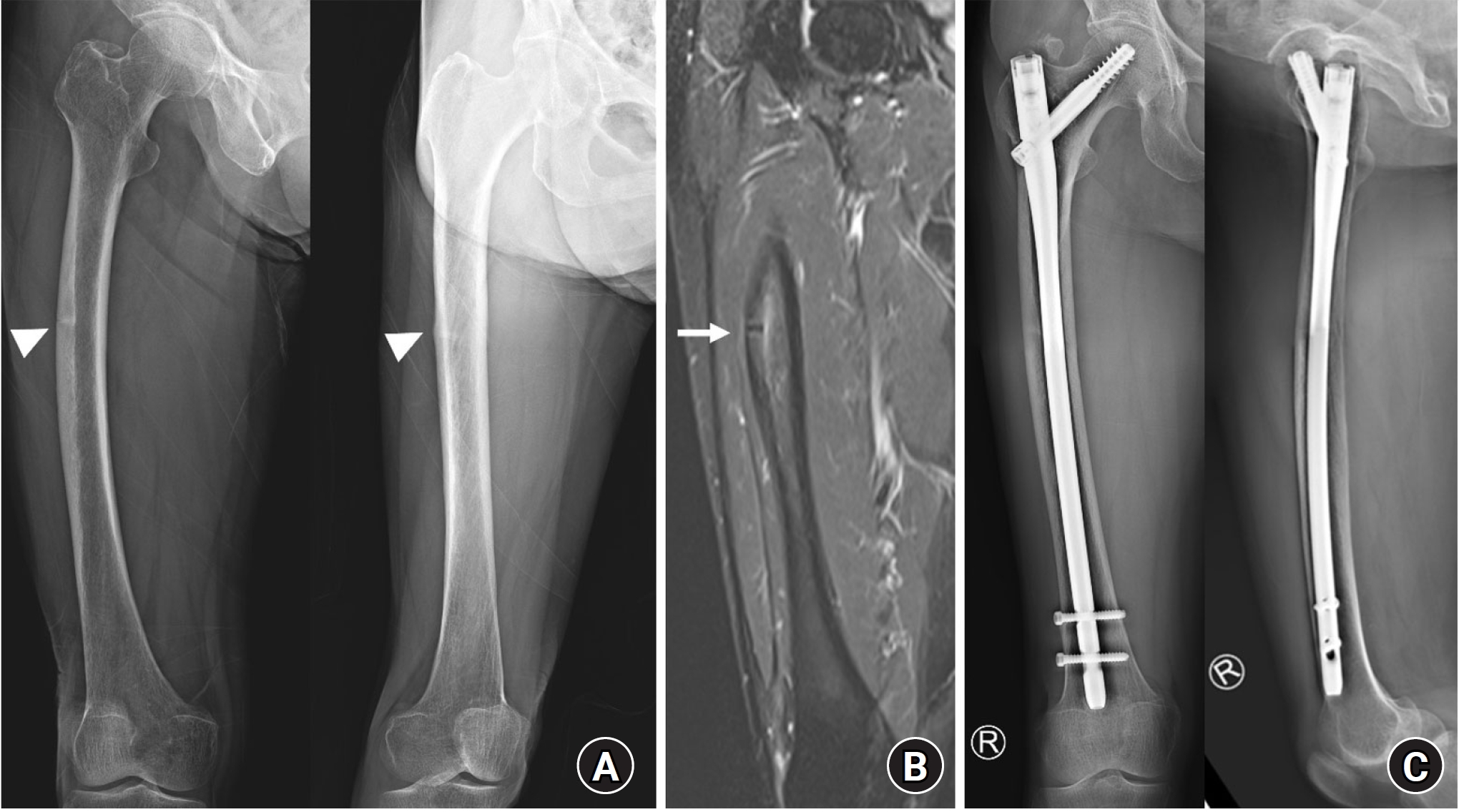
Fig. 5.An 80-year-old female patient with an 8-year history of alendronate use and intermittent left thigh pain underwent dual-energy X-ray absorptiometry (DXA) as part of her annual medical checkup. The DXA image revealed lateral cortical beaking (arrow). Further evaluation with a radiograph showed similar findings, including a black line (arrowhead), indicating an incomplete atypical femoral fracture.
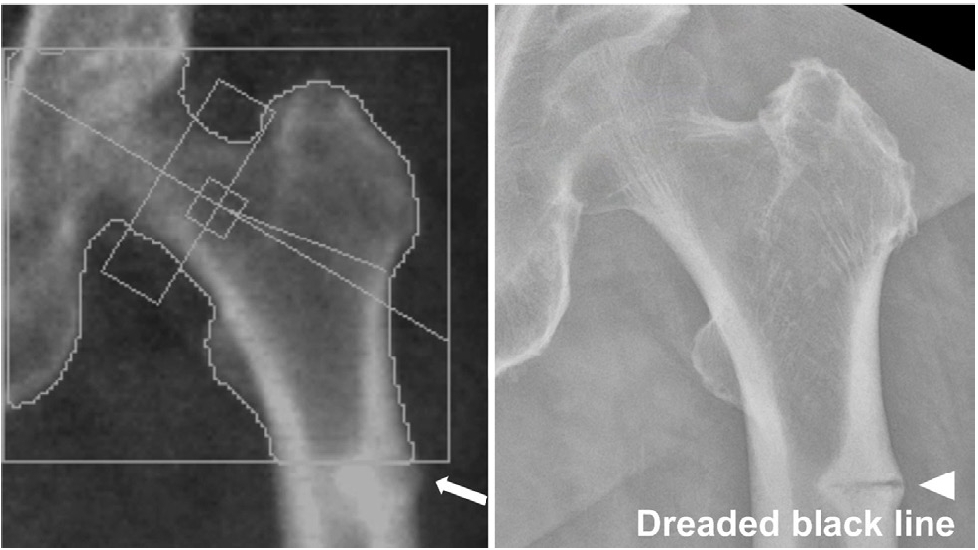
Table 1.Revised case definition of AFF from the ASBMR Task Force (2013)
|
Definition |
|
Pre-requisites |
Fracture location must be located along the femoral diaphysis from just distal to the lesser trochanter to just proximal to the supracondylar flare. |
|
In addition, at least four of five major features must be present. |
|
No minor features are required but have sometimes been associated with these fractures. |
|
Major criteria |
1. The fracture is associated with minimal or no trauma, as in a fall from a standing height or less. |
|
2. The fracture line originates at the lateral cortex and is transverse, although it may become oblique as it progresses medially. |
|
3. Complete fractures extend through both cortices and may be associated with a medial spike; incomplete fractures involve only the lateral cortex. |
|
4. The fracture is non- or minimally comminuted. |
|
5. Localized periosteal or endosteal thickening of the lateral cortex is present at the fracture site (“beaking” or “flaring”). |
|
Minor criteria |
1. Generalized increase in the cortical thickness of the femoral diaphysis. |
|
2. Unilateral or bilateral prodromal symptoms, such as dull or aching pain in the groin or thigh. |
|
3. Bilateral incomplete or complete femoral diaphysis fractures. |
|
4. Delayed fracture healing. |
References
- 1. Wells GA, Hsieh SC, Peterson J, et al. Alendronate for the primary and secondary prevention of osteoporotic fractures in postmenopausal women. Cochrane Database Syst Rev 2025;1:CD001155.ArticlePubMed
- 2. Geissler JR, Bajaj D, Fritton JC. American Society of Biomechanics Journal of Biomechanics Award 2013: cortical bone tissue mechanical quality and biological mechanisms possibly underlying atypical fractures. J Biomech 2015;48:883-94.ArticlePubMedPMC
- 3. Kharwadkar N, Mayne B, Lawrence JE, Khanduja V. Bisphosphonates and atypical subtrochanteric fractures of the femur. Bone Joint Res 2017;6:144-53.ArticlePubMedPMCPDF
- 4. Gedmintas L, Solomon DH, Kim SC. Bisphosphonates and risk of subtrochanteric, femoral shaft, and atypical femur fracture: a systematic review and meta-analysis. J Bone Miner Res 2013;28:1729-37.ArticlePubMedPDF
- 5. Kang JS, Won YY, Kim JO, et al. Atypical femoral fractures after anti-osteoporotic medication: a Korean multicenter study. Int Orthop 2014;38:1247-53.ArticlePubMedPMCPDF
- 6. Nho JH, Jang BW, Lee DW, et al. Position statement: atypical femoral fracture from the Korean Society for Bone and Mineral Research in 2023. J Bone Metab 2023;30:209-17.ArticlePubMedPMCPDF
- 7. Lim SJ, Yeo I, Yoon PW, et al. Incidence, risk factors, and fracture healing of atypical femoral fractures: a multicenter case-control study. Osteoporos Int 2018;29:2427-35.ArticlePubMedPDF
- 8. Yang KH, Min BW, Ha YC. Atypical femoral fracture: 2015 position statement of the Korean Society for Bone and Mineral Research. J Bone Metab 2015;22:87-91.ArticlePubMedPMC
- 9. Collins L, Ronan A, Hutcheon E, Ebeling PR, Grill V, Nguyen HH. Atypical fractures at non-classical sites associated with anti-resorptive therapy: a systematic review. J Bone Miner Res 2024;39:1722-34.ArticlePubMedPMCPDF
- 10. Auger K, Lee J, Hong IS, Jankowski JM, Liporace FA, Yoon RS. Bilateral atypical femoral fracture in a bisphosphonate-naïve patient with prior long-term denosumab therapy: a case report of the management strategy and a literature review. J Clin Med 2024;13:2785.ArticlePubMedPMC
- 11. Byun SE, Lee KJ, Shin WC, Moon NH, Kim CH. The effect of teriparatide on fracture healing after atypical femoral fracture: a systematic review and meta-analysis. Osteoporos Int 2023;34:1323-34.ArticlePubMedPDF
- 12. Salamah HM, Abualkhair KA, Kamal SK, et al. The effect of teriparatide on patients with atypical femur fractures: a systematic review and meta-analysis. Arch Orthop Trauma Surg 2024;144:1091-106.ArticlePubMedPDF
- 13. Song G, Jeong Y, Nam WD, Kim KH. Teriparatide does not have beneficial effects on bone healing in complete atypical femur fractures. Calcif Tissue Int 2024;115:169-73.ArticlePubMedPDF
- 14. Akagawa M, Miyakoshi N, Tsuchie H, et al. Successful treatment of atypical femoral fracture with bowed femur using contralateral intramedullary nail combined with early daily teriparatide. Acta Med Okayama 2022;76:333-8.PubMed
- 15. Coscia A, Wilson J, Muscatelli S, Hake M. Operative management of atypical femur fractures. J Orthop Trauma 2024;38(8S):S7-8.Article
- 16. Kim KH, Song G. Intramedullary nailing for atypical femoral fracture with lateral bowing: does medial gap matter? Geriatr Orthop Surg Rehabil 2022;13:21514593211070130.ArticlePubMedPMCPDF
- 17. Shim BJ, Won H, Kim SY, Baek SH. Surgical strategy of the treatment of atypical femoral fractures. World J Orthop 2023;14:302-11.ArticlePubMedPMC
- 18. Shane E, Burr D, Abrahamsen B, et al. Atypical subtrochanteric and diaphyseal femoral fractures: second report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res 2014;29:1-23.PubMed
- 19. Adams AL, Xue F, Chantra JQ, et al. Sensitivity and specificity of radiographic characteristics in atypical femoral fractures. Osteoporos Int 2017;28:413-7.ArticlePubMedPDF
- 20. Odvina CV, Zerwekh JE, Rao DS, Maalouf N, Gottschalk FA, Pak CY, et al. a potential complication of alendronate therapy. J Clin Endocrinol Metab 2005;90:1294-301.PubMed
- 21. Eastell R, Lang T, Boonen S, et al. Effect of once-yearly zoledronic acid on the spine and hip as measured by quantitative computed tomography: results of the HORIZON Pivotal Fracture Trial. Osteoporos Int 2010;21:1277-85.ArticlePubMedPDF
- 22. Kim SY, Schneeweiss S, Katz JN, Levin R, Solomon DH. Oral bisphosphonates and risk of subtrochanteric or diaphyseal femur fractures in a population-based cohort. J Bone Miner Res 2011;26:993-1001.ArticlePubMedPDF
- 23. Black DM, Kelly MP, Genant HK, et al. Bisphosphonates and fractures of the subtrochanteric or diaphyseal femur. N Engl J Med 2010;362:1761-71.ArticlePubMed
- 24. Shane E, Burr D, Ebeling PR, et al. Atypical subtrochanteric and diaphyseal femoral fractures: report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res 2010;25:2267-94.PubMed
- 25. Girgis CM, Seibel MJ. Population and treatment-based incidence estimates of atypical fractures. Med J Aust 2011;194:666.ArticlePDF
- 26. Dell R, Greene D. A proposal for an atypical femur fracture treatment and prevention clinical practice guideline. Osteoporos Int 2018;29:1277-83.ArticlePubMedPDF
- 27. Lo JC, Hui RL, Grimsrud CD, et al. The association of race/ethnicity and risk of atypical femur fracture among older women receiving oral bisphosphonate therapy. Bone 2016;85:142-7.ArticlePMC
- 28. Bottai V, Antognetti D, Troiano E, et al. Prevalence of atypical femoral fractures, a clinical update: a comparative retrospective study 7 years later. Injury 2024;55 Suppl 4:111346.ArticlePubMed
- 29. Lee YK, Kim TY, Ha YC, et al. Atypical subtrochanteric fractures in Korean hip fracture study. Osteoporos Int 2017;28:2853-8.ArticlePubMedPDF
- 30. Khan SA, Kanis JA, Vasikaran S, et al. Elimination and biochemical responses to intravenous alendronate in postmenopausal osteoporosis. J Bone Miner Res 1997;12:1700-7.ArticlePubMedPDF
- 31. Gertz BJ, Holland SD, Kline WF, Matuszewski BK, Porras AG. Clinical pharmacology of alendronate sodium. Osteoporos Int 1993;3 Suppl 3:S13-6.ArticlePubMedPDF
- 32. Ahn J, Kim CH, Kim JW. Comparison of bone turnover suppression in atypical femoral fractures and osteoporotic hip fractures. Sci Rep 2024;14:19974.ArticlePubMedPMCPDF
- 33. Black DM, Geiger EJ, Eastell R, et al. Atypical femur fracture risk versus fragility fracture prevention with bisphosphonates. N Engl J Med 2020;383:743-53.ArticlePubMedPMC
- 34. Zanchetta MB, Diehl M, Buttazzoni M, et al. Assessment of bone microarchitecture in postmenopausal women on long-term bisphosphonate therapy with atypical fractures of the femur. J Bone Miner Res 2014;29:999-1004.ArticlePubMedPDF
- 35. Somford MP, van Ruijven LJ, Kloen P, Bakker AD. Histological and micro Computed Tomography analysis of a femoral stress fracture associated with prolonged bisphosphonate use. Clin Cases Miner Bone Metab 2017;14:92-6.ArticlePubMedPMC
- 36. Martelli S, Pivonka P, Ebeling PR. Implications for atypical femoral fractures. Clin Biomech (Bristol) 2014;29:869-76.PubMed
- 37. Haider IT, Schneider P, Michalski A, Edwards WB. Influence of geometry on proximal femoral shaft strains: implications for atypical femoral fracture. Bone 2018;110:295-303.ArticlePubMed
- 38. Oh Y, Fujita K, Wakabayashi Y, Kurosa Y, Okawa A. Location of atypical femoral fracture can be determined by tensile stress distribution influenced by femoral bowing and neck-shaft angle: a CT-based nonlinear finite element analysis model for the assessment of femoral shaft loading stress. Injury 2017;48:2736-43.ArticlePubMed
- 39. Hyodo K, Nishino T, Kamada H, Nozawa D, Mishima H, Yamazaki M., the characteristics of patients with atypical femoral fractures. Location of fractures and the characteristics of patients with atypical femoral fractures: analyses of 38 Japanese cases. J Bone Miner Metab 2017;35:209-14.ArticlePubMedPDF
- 40. Hagen JE, Miller AN, Ott SM, et al. Association of atypical femoral fractures with bisphosphonate use by patients with varus hip geometry. J Bone Joint Surg Am 2014;96:1905-9.ArticlePubMed
- 41. Koh JS, Goh SK, Png MA, Ng AC, Howe TS. Distribution of atypical fractures and cortical stress lesions in the femur: implications on pathophysiology. Singapore Med J 2011;52:77-80.PubMed
- 42. Jang SP, Yeo I, So SY, et al. Atypical femoral shaft fractures in female bisphosphonate users were associated with an increased anterolateral femoral bow and a thicker lateral cortex: a case-control study. Biomed Res Int 2017;2017:5932496.ArticlePubMedPMCPDF
- 43. Shin WC, Moon NH, Jang JH, Park KY, Suh KT. Anterolateral femoral bowing and loss of thigh muscle are associated with occurrence of atypical femoral fracture: effect of failed tension band mechanism in mid-thigh. J Orthop Sci 2017;22:99-104.ArticlePubMed
- 44. Morin SN, Wall M, Belzile EL, et al. Assessment of femur geometrical parameters using EOS™ imaging technology in patients with atypical femur fractures; preliminary results. Bone 2016;83:184-9.ArticlePubMed
- 45. Chen LP, Chang TK, Huang TY, Kwok TG, Lu YC. The correlation between lateral bowing angle of the femur and the location of atypical femur fractures. Calcif Tissue Int 2014;95:240-7.ArticlePubMedPDF
- 46. Saita Y, Ishijima M, Mogami A, et al. The fracture sites of atypical femoral fractures are associated with the weight-bearing lower limb alignment. Bone 2014;66:105-10.ArticlePubMed
- 47. Jung IJ, Kim JW. Differences in femur geometry and bone markers in atypical femur fractures and the general population. Sci Rep 2021;11:24149.ArticlePubMedPMCPDF
- 48. Hwang D, Kim CH, Lee Y, Kim JW. Association of atypical femoral fracture location and lower limb mechanical axis: a computed tomography-based finite element analysis. Osteoporos Int 2022;33:1285-93.ArticlePubMedPDF
- 49. Del Real Á, Cruz R, Sañudo C, et al. High Frequencies of genetic variants in patients with atypical femoral fractures. Int J Mol Sci 2024;25:2321.ArticlePubMedPMC
- 50. Ellacott M, Bilgehan Çevik H, Giannoudis PV. Is there genetic susceptibility for atypical femoral fractures? Injury 2024;55:111312.ArticlePubMed
- 51. Zhou W, Ås J, Shore-Lorenti C, et al. Gene-based association analysis of a large patient cohort provides insights into genetics of atypical femur fractures. J Bone Miner Res 2024;39:1315-26.ArticlePubMedPMCPDF
- 52. Marini F, Giusti F, Marasco E, et al. High frequency of heterozygous rare variants of the SLC34A1 and SLC9A3R1 genes in patients with atypical femur fracture. Eur J Endocrinol 2023;188:lvad001.ArticlePubMedPDF
- 53. Zhou W, van Rooij JG, van de Laarschot DM, et al. Prevalence of monogenic bone disorders in a dutch cohort of atypical femur fracture patients. J Bone Miner Res 2023;38:896-906.PubMed
- 54. Garcia-Giralt N, Ovejero D, Grinberg D, et al. Assessing the contribution of genes involved in monogenic bone disorders to the etiology of atypical femoral fractures. Hum Genomics 2024;18:87.ArticlePubMedPMCPDF
- 55. Selga J, Nuñez JH, Minguell J, Lalanza M, Garrido M. Simultaneous bilateral atypical femoral fracture in a patient receiving denosumab: case report and literature review. Osteoporos Int 2016;27:827-32.ArticlePubMedPDF
- 56. Bone HG, Wagman RB, Brandi ML, et al. 10 Years of denosumab treatment in postmenopausal women with osteoporosis: results from the phase 3 randomised FREEDOM trial and open-label extension. Lancet Diabetes Endocrinol 2017;5:513-23.PubMed
- 57. Fitzpatrick D, Seebah S, Lannon R, Mc Carroll K. Atypical femoral fracture after denosumab for the treatment of osteoporosis in bisphosphonate naïve patients: two further cases. Osteoporos Int 2023;34:1807-8.ArticlePubMedPDF
- 58. Kumar S, Chang R, Reyes M, Diamond T. Atypical femoral fracture in a bisphosphonate-naïve patient on denosumab for osteoporosis. Arch Osteoporos 2022;17:131.ArticlePubMedPDF
- 59. Leclerc JT, Michou L, Vaillancourt F, Pelet S, Simonyan D, Belzile EL. Prevalence and characteristics of atypical periprosthetic femoral fractures. J Bone Miner Res 2019;34:83-92.ArticlePubMedPDF
- 60. Park YC, Song HK, Zheng XL, Yang KH. Intramedullary nailing for atypical femoral fracture with excessive anterolateral bowing. J Bone Joint Surg Am 2017;99:726-35.ArticlePubMed
- 61. Lee YK, Park CH, Kim KC, Hong SH, Ha YC, Koo KH. Frequency and associated factor of atypical periprosthetic femoral fracture after hip arthroplasty. Injury 2018;49:2264-8.ArticlePubMed
- 62. Kim JW, Oh CW, Park KH, Oh JK, Yoon YC, Kim JK. Peri-implant atypical femoral fracture after nail or plate osteosynthesis. J Orthop Sci 2022;27:866-75.ArticlePubMed
- 63. Ferreira P, Bates P, Daoub A, Dass D. Is bisphosphonate use a risk factor for atypical periprosthetic/peri-implant fractures?: a metanalysis of retrospective cohort studies and systematic review of the current evidence. Orthop Traumatol Surg Res 2023;109:103475.ArticlePubMed
- 64. Hashimoto J, Oh Y, Yamamoto K, et al. Frequency and pathogenesis of periprosthetic atypical femoral fractures associated with total knee arthroplasty: a multicenter prospective study with complementary histopathological and biomechanical analysis. Injury 2024;55:111136.ArticlePubMed
- 65. Kimura S, Sunouchi T, Watanabe S, et al. Latent metabolic bone disease, skeletal dysplasia and other conditions related to low bone formation among 38 patients with subtrochanteric femoral fractures: a retrospective observational study. Osteoporos Int 2024;35:1633-43.ArticlePubMedPMCPDF
- 66. Dhanekula ND, Crouch G, Byth K, et al. Asian ethnicity and femoral geometry in atypical femur fractures: independent or interdependent risk factors? JBMR Plus 2022;6:e10607. ArticlePubMedPMCPDF
- 67. Koh JH, Myong JP, Yoo J, et al. Predisposing factors associated with atypical femur fracture among postmenopausal Korean women receiving bisphosphonate therapy: 8 years’ experience in a single center. Osteoporos Int 2017;28:3251-9.ArticlePubMedPDF
- 68. Sato H, Kondo N, Kurosawa Y, et al. Incidence and clinical course of femoral localized periosteal thickening and atypical femoral fracture over a 10-year period in patients with autoimmune inflammatory rheumatic disease. JBMR Plus 2024;8:ziae090.ArticlePubMedPMCPDF
- 69. Kharazmi M, Michaëlsson K, Hallberg P. Prodromal symptoms in patients with bisphosphonate-associated atypical fractures of the femur. J Bone Miner Metab 2015;33:516-22.ArticlePubMedPDF
- 70. Schneider PS, Wall M, Brown JP, Cheung AM, Harvey EJ, Morin SN. Atypical femur fractures: a survey of current practices in orthopedic surgery. Osteoporos Int 2017;28:3271-6.ArticlePubMedPDF
- 71. Dell RM, Adams AL, Greene DF, et al. Incidence of atypical nontraumatic diaphyseal fractures of the femur. J Bone Miner Res 2012;27:2544-50.ArticlePubMedPDF
- 72. Tins BJ, Garton M, Cassar-Pullicino VN, Tyrrell PN, Lalam R, Singh J. Stress fracture of the pelvis and lower limbs including atypical femoral fractures: a review. Insights Imaging 2015;6:97-110.ArticlePubMedPDF
- 73. Png MA, Koh JS, Goh SK, Fook-Chong S, Howe TS. Bisphosphonate-related femoral periosteal stress reactions: scoring system based on radiographic and MRI findings. AJR Am J Roentgenol 2012;198:869-77.ArticlePubMed
- 74. Kim S, Yang KH, Lim H, et al. Detection of prefracture hip lesions in atypical subtrochanteric fracture with dual-energy x-ray absorptiometry images. Radiology 2014;270:487-95.ArticlePubMed
- 75. Cheung AM, McKenna MJ, van de Laarschot DM, et al. Detection of atypical femur fractures. J Clin Densitom 2019;22:506-16.ArticlePubMed
- 76. Smith D, Knapp PK, Wright DC, Hollick DR. Dual Energy X-Ray Absorptiometry (DXA) extended femur scans to support opportunistic screening for incomplete atypical femoral fractures: a short term in-vivo precision study. J Clin Densitom 2023;26:101352.ArticlePubMed
- 77. Cha SM, Kim YK, Shin HD, Park JY, Lee SH. Importance of whole-body scintigraphy or positron emission tomography for early detection of femoral lesions in breast cancer patients treated with bisphosphonates. Clin Orthop Surg 2023;15:659-67.ArticlePubMedPMCPDF
 , Jeong-Hyun Koh, MD
, Jeong-Hyun Koh, MD , Seungyeob Sakong, MD
, Seungyeob Sakong, MD , Jung-Taek Kim, MD, PhD
, Jung-Taek Kim, MD, PhD










 E-submission
E-submission KOTA
KOTA TOTA
TOTA TOTS
TOTS
 ePub Link
ePub Link Cite
Cite